
Noble gases, which constitute the left-most group of the periodic table and include elements such as Neon or Argon are characterized by their chemical inertness. Nonmetals can be further divided into two categories: noble gases and reactive nonmetals. Non-metals generally have low melting and boiling points, are usually brittle at room temperatures, are poor conductors of heat and electricity, and typically enter into covalent or ionic bonds with other elements. In simplest terms, a nonmetal on the periodic table is an element that mostly lacks any metallic properties. Depending on which specific properties of metals are of interest to the researcher, they will classify them into different subgroups. The category of metals can be further divided into ferrous (magnetic) and non-ferrous metals, Alkali, transition, and base metals, and so forth. The chemical nature of metallic bonds explains many of the unique properties of metals, such as their strength, durability, luster, and conductive properties.

In other words, a metal compound can be thought of as a positively charged lattice of cations that shares electrons freely across its structure. When metal elements form compounds, their bonds are characterized by the delocalization of electron pairs. Metals represent the largest classification of element-type as approximately 95 of the 118 discovered elements are metals. MetalsĮlements classified as metals are unified by their tendency to form cations, enter into metallic chemical bonds with other metals, are prone to oxidization, and are generally good conductors of heat and electricity. These fuzzy boundaries merely reflect the fact that there is no universal agreement about which elements fall within these categories, but in general, scientists work with this general understanding of the division of element-types. In other cases, arsenic is classified as a metalloid as it is brittle at room temperature and readily dissolves in water. For example, in some cases, arsenic would be classified as a metal due to its strong conductive properties. Although these categories are commonly used, classifications at the edge have somewhat fuzzy boundaries.
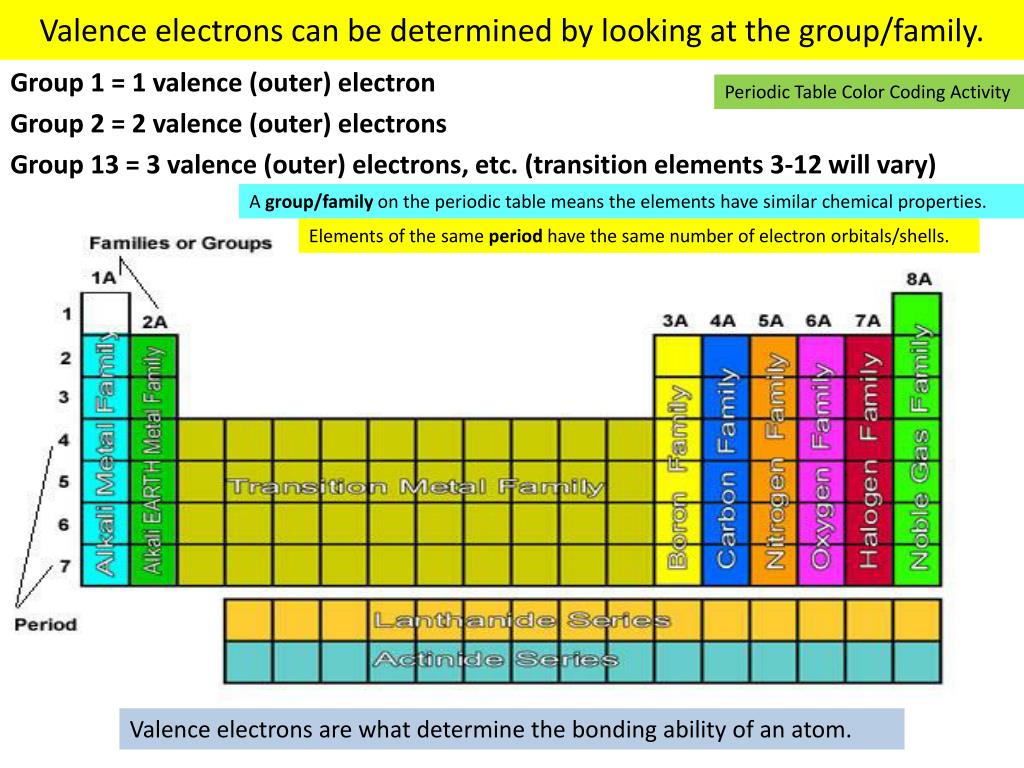
In general, chemists divide the elements into three major types: metals (purple) non-metals (blue) and metalloids (green). In the above photo, the elements are color-coded respective to the general type of substance that element falls under. “Simple Periodic Table Chart-en” by Offnfopt via Wikimedia Commons is licensed under CC0 Color-Coded Periodic Table By Element Type Now that we have some basics of the periodic table under our belt, let’s move on to specific ways to organize the chemical elements. Similarly, both fluorine and chlorine are halogens and are characterized by their tendency to spontaneously form strong ionic bonds with electronegatively weak elements. Both carbon and silicon have 4 open valence electrons, which explains why they exhibit similar chemical properties. For example, carbon is in the same group as silicon as they both exhibit similar chemical properties. The result is the elements that are arranged in the same groups share similar chemical properties. These periodic changes correspond to periodic changes in the chemical behavior of elements as you go along the row. “What the ocean was to a child, the periodic table is to the chemist.” - Karl Barry Sharpless For example, elements on the leftmost part of the rows have few exterior electrons in their outer shells, and the number increases as you go from left to right. Elements in arranged in a period exhibit recurring chemical properties as you go from left to right. The periodic table is organized in a matrix of horizontal rows and vertical columns called periods and groups, respectively. Each cell in the periodic table has a symbol for the element, the atomic number of the element which corresponds to the number of protons in an atom of that element, and the average atomic weight of that element. What Is The Periodic Table?įirst some general facts about the periodic table. One of the most common ways of grouping elements in the periodic table are groupings based on general element-type, such as by the categories, metal, nonmetal, and metalloid. Because there are a number of ways to classify the elements, there exist a number of color-coding schemes we could use to point out specific properties that groups of elements share in common. Elements in the periodic table can be color-coded together according to atomic number, electron configuration, and recurring chemical properties. The color-coded periodic table is a tabular representation of the different chemical elements.
